Acute traumatic injuries : absolute emergencies
- ACUTE SPINAL CORD INJURIES : a rare but dreadful condition
30.000 new cases per year in USA+EU; no treatment; standard of care : decompressive surgery;
Victims are mostly: young males (traffic and sport accidents) + the elderly ones (falls).
Long-term handicaps include paraplegia and tetraplegia.
- TRAUMATIC BRAIN INJURIES : an exceedingly frequent condition
5-7 million new cases per year; no treatment; standard of care: none;
Victims are mostly : kids and males (sport, violence, traffic, wars, work accidents);
long-term disabilities include depression, memory deficits, higher prevalence of neurological Disorders (Parkinson, dementias).
- STROKE : stroke strikes every 4 minute
3-5 million new cases per year; consists in sudden ischemia (blot clots, 80% cases) or hemorraghia ;
Most frequent in the elderly but fast growing in younger people;
Removal of blood clot is possible within the first 4h post-trauma (benefits to less than 20% patients)
In any case : neuronal loss is irreversible.
Act fast to limit secondary damages
Spinal Cord injuries, Traumatic Brain Injuries and Strokes, a cascade of damages
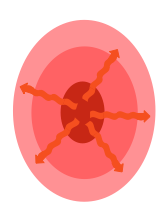
EPICENTER
The initial trauma called the initial lesion, is focal and/or diffuse:
Compression
Contusion
Stretching
Distortion
Laceration
Hematoma
Hypoxia
WAVES OF DEATH
Leads to cascades of damages called secondary lesions that spread from initial lesion:Neuronal suffering and death
Our positioning : an emergency injection of MAP4343 to prevent secondary damages after the initial lesion.
MAP4343 promotes rapid and Impressive recovery after acute traumatic injuries
Studies supported by OSEO and by the US Army
In 3 different animal models of Spinal Cord Injuries, MAP4343:
- Promotes motor recovery (BBB scores)
- Promotes axonal growth
In 3 different animal models of Traumatic Brain Injuries, MAP4343:
- Decreases edema (MRI studies)
- Decreases anxiety
- Promotes synaptic remodelling
- Protects Hippocampus
In the most widely used animal model of Stroke, MAP4343:
- Promotes rapid motor recovery (foot fault, rotarod & neurological score)
An emergency injection of MAP4343 could prevent secondary damages and help neurons to regenerate, while helping general rehabilitation and mood thanks of its antidepressant properties.
.
CHU Bicêtre, Batiment Pincus, 80 rue du général Leclerc, 94270 Le Kremlin-Bicêtre, France.
- (telephone:)
+33 (0)1 49 59 18 71 - (email:)
administration@mapreg.com